Molecular Neuroscience
TEL +81-6-6879-3661
FAX +81-6-6879-3669
Overview
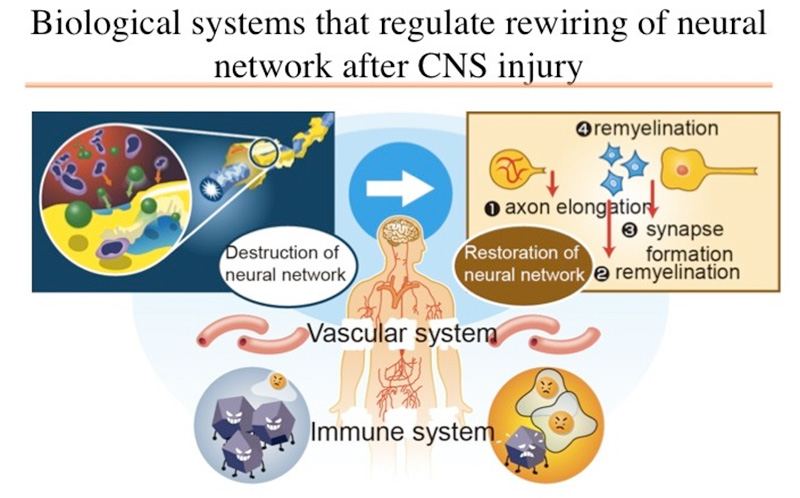
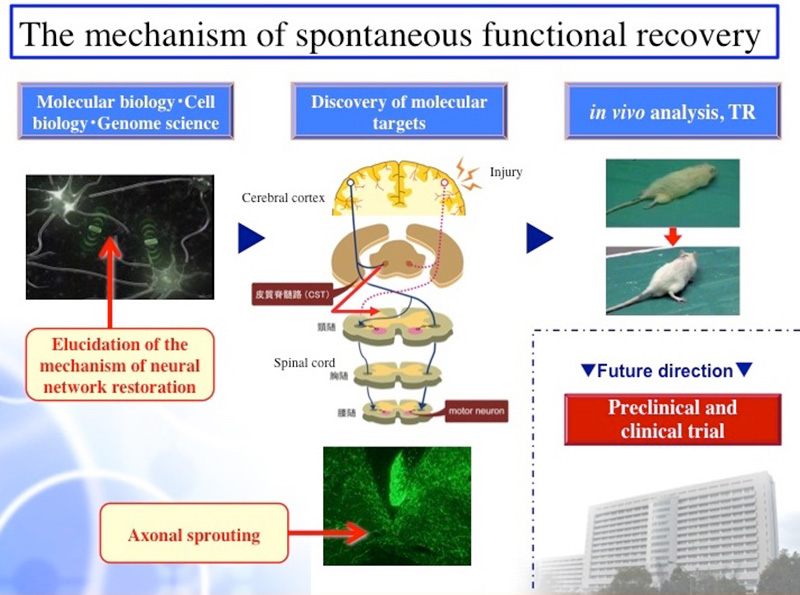
Disorders of the central nervous system, such as cerebrovascular diseases, cerebrospinal trauma, and encephalomyelitis, often cause spatiotemporal changes in the nervous system and in various biological systems, such as the immune system and vascular system. We have analyzed disorders of the neural networks in the central nervous system and the subsequent restoration process from the perspective of the functional network of biological systems (Fig. 1 and 2). Further, we have analyzed the mechanism by which the spatiotemporal dynamics in those biological systems control a series of processes. Particularly, the ultimate goal of this study is to elucidate the control mechanism exerted by the associations among the nervous system, immune system, and vascular system. Additionally, we aim to elucidate the principles involved in the operation of living organisms with neural network disorders within the central nervous system by observing such disorders and their functional recovery process with respect to the dynamics of the entire biological system and by conducting a comprehensive analysis of the association between each system. We perceive the central nervous system as a single organ within a biological system; further, studies from the perspective of the involvement of the entire biological system in disorders and recovery of neural networks are scarce. By perceiving disorders in the neural networks and the biological reactions during the subsequent recovery process as a “scrap-and-build” strategy, we aim to elucidate the mechanisms behind a series of reactions as well as their significance that may potentially create a new and never-seen-before trend in Life Sciences.
Principal Investigator
Toshihide Yamashita Professor

Research field
Molecular Mechanisms Regulating the Regeneration and Plasticity of Injured Central Nervous System
Education history
| 1990.3 | M.D. (Osaka University) |
|---|---|
| 1997 | Ph.D. (Medical Science, Osaka University) |
Research and career history
| 1990 | Medical Staff in Department of Neurosurgery, Osaka University Hospital (-94) |
|---|---|
| 1996 | Assistant Professor, Department of Anatomy and Neuroscience, Graduate School of Medicine, Osaka University (-98) |
| 1998 | Research Fellow, Department of Neurobiochemistry, Max-Planck Institute of Neurobiology (-2000) |
| 2001 | Associate Professor, Department of Anatomy and Neuroscience, Graduate School of Medicine, Osaka University (-2003.10) |
| 2003.11 | Professor and Chairman, Department of Neurobiology, Graduate School of Medicine, Chiba University (-2007.11) |
| 2007.12 | Professor and Chairman, Department of Molecular Neuroscience, Graduate School of Medicine, Osaka University (-Present) |
| 2009.4 | Professor (Additional), Graduate School of Frontier Biosciences, Osaka University)(-Present) |
| 2017.4 | PI (Additional), WPI Immunology Frontier Research Center (-Present) |
| 2017.4 | Osaka University Distinguished Professor |
| 2018.7 | Professor, Department of Neuro-Medical Science, Graduate School of Medicine, Osaka University |
Prize
| 2005 | Ameritec Prize (Ameritec Foundation, USA) |
|---|---|
| 2010 | Japan Society for the Promotion of Science Prize (Japan) |
| 2011 | Osaka Science Prize (Japan) |
| 2014 | Commendation for Science and Technology by the Minister of Education, Culture, Sports, Science and Technology |
Members
- Toshihide Yamashita Professor
yamashitamolneu.med.osaka-u.ac.jp - Takahide Itokazu Professor
t.itokazumolneu.med.osaka-u.ac.jp - Nao Shibuya Associate Professor (Lecturer)
nshibuyamolneu.med.osaka-u.ac.jp
Achievements
Publications
- Fujita, Y., Masuda, K., Nakato, R., Katou, Y., Tanaka, T., Nakayama, M., Takao, K., Miyakawa, T., Shirahige, K. and Yamashita, T. (2017) Cohesin regulates formation of neuronal networks in the brain. J. Exp. Med. 214, 1431-1452.
- Fujitani, M., Zhang, S., Fujiki, R., Fujihara, Y. and Yamashita, T. (2017) A chromosome 16p13.11 microduplication causes hyperactivity through dysregulation of miR-484/protocadherin-19 signaling. Mol. Psychiatry 22, 364-374.
- Siebold, C., Yamashita, T., Monnier, P., Mueller, B.K. and Pasterkamp, R.J. (2017) RGMs: Structural Insights, Molecular Regulation and Downstream Signaling. Trends Cell Biol. 27, 365-378.
- Hayano, Y., Takasu, K., Koyama, Y., Ogawa, K., Minami, K., Asaki, T., Kitada, K., Kuwabara, S. and Yamashita, T. (2016) Dorsal horn interneuron-derived Netrin-4 contributes to spinal sensitization in chronic pain via Unc5B. J. Exp. Med. 213, 2949-2966
- van Erp, S., van den Heuvel, D.M.A., Fujita, Y., Robinson, R.A., Hellemons, A.J., Adolfs, Y., Van Battum, E.Y., Blokhuis, A.M., Kuijpers, M., Demmers, J., Hedman, H., Hoogenraad, C.C., Siebold, C., Yamashita, T. and Pasterkamp, R.J. (2015) Lrig2 negatively regulates ectodomain shedding of axon guidance receptors by ADAM proteases. Dev. Cell 35, 537-552.
- Arima, Y., Kamimura, D., Yamada, M., Sabharwal, L., Harada, M., Kawamoto, T., Yamashita, T. Ogura, H., Hirano, T. and Murakami, M. (2015) Pain-mediated sympathetic activation induces relapse via microglia accumulation in a multiple sclerosis model. eLife 4, e08733.
- Tanabe, S. and Yamashita, T. (2014) Repulsive guidance molecule-a is involved in Th17 cell-induced neurodegeneration in autoimmune encephalomyelitis. Cell Rep. 9, 1459-1470.
- Ueno, M., Fujiki, R. and Yamashita, T. (2014) A selector orchestrates cortical function. Nat. Neurosci. 17, 1016-1017.
- Hayano, Y., Sasaki, K., Ohmura, N., Takemoto, M., Maeda, Y., Yamashita, T.,Hata, Y., Kitada, K and Yamamoto, N. (2014) Netrin-4 regulates thalamocortical axon branching in an activity-dependent fashion. Proc. Natl. Acad. Sci. USA 111, 15226-15231.
- Ueno, M. and Yamashita, T. (2014) Bidirectional tuning of microglia in the developing brain: From neurogenesis to neural circuit formation. Curr. Opin. Neurobiol. 27C, 8-15.
- Ueno, M., Fujita, Y., Tanaka, T., Nakamura, Y., Kikuta, J., Ishii, M. and Yamashita, T. (2013) Layer V cortical neurons require microglial support for survival during postnatal development. Nat. Neurosci. 16, 543-551.
- Muramatsu, R., Takahashi, C., Miyake, S., Fujimura, H., Mochizuki, H. and Yamashita, T. (2012) Angiogenesis induced by CNS inflammation promotes neural remodeling through vessel-derived prostacyclin. Nature Medicine 18, 1658-1664.
- Ueno, M., Hayano, Y., Nakagawa, H. and Yamashita, T. (2012) Intraspinal rewiring of the corticospinal tract requires target-derived brain-derived neurotrophic factor and compensates lost function after brain injury. Brain 135, 1253-1267.
- Muramatsu, R., Kubo, T., Mori, M., Nakamura, Y., Fujita, Y., Akutsu, T., Okuno, T., Taniguchi, J., Kumanogoh, A., Yoshida, M., Mochizuki, H., Kuwabara, S. and Yamashita, T. (2011) RGMa modulates T cell responses and is involved in autoimmune encephalomyelitis. Nature Medicine 17, 488-494.
- Fujita, Y., Endo, S., Takai, T. and Yamashita, T. (2011) Myelin suppresses axon regeneration by PIR-B/SHP-mediated inhibition of Trk activity. EMBO J. 30, 1389-1401.
- Sanuki, R., Onishi, A., Koike, C., Muramatsu, R., Watanabe, S., Muranishi, Y, Irie, S., Ueno, S., Koyasu, T., Matsui, R., Cherasse, Y., Urade, Y., Watanabe, D., Kondo, M., Yamashita, T. and Furukawa, T. (2011) miR-124a is required for hippocampal axogenesis and retinal cone survival through Lhx2 suppression. Nat. Neurosci. 14, 1125–1134
- Yamagishi, S., Hampe1, F., Hata, K., del Toro, D., Schwark, M., Kvachnina, E., Bastmeyer, M., Yamashita, T., Tarabykin, V., Klein, R. and Egea. J. (2011) FLRT2 and FLRT3 act as repulsive guidance cues for Unc5-positive neurons. EMBO J. 30, 2920-2933.
- Hata, K., Kaibuchi, K., Inagaki, S. and Yamashita, T. (2009) Unc5B associates with LARG to mediate the action of repulsive guidance molecule. J. Cell Biol. 184, 737-750.
- Hata, K., Fujitani, M., Yasuda, Y., Doya, H., Saito, T., Yamagishi, S., Mueller, B.K. and Yamashita, T. (2006) RGMa inhibition promotes axonal growth and recovery after spinal cord injury. J. Cell Biol. 173, 47-58.
- Yamashita, T. & Tohyama, M. (2003) The p75 receptor acts as a displacement factor that releases Rho from Rho GDI. Nature Neurosci. 6, 461-467.
- Higuchi, H., Yamashita, T., Yoshikawa, H. & Tohyama, M. (2003) PKA phosphorylates the p75 receptor and regulates its localization to lipid rafts. EMBO J. 22, 1790-1800.
- Yamashita, T., Higuchi, H., and Tohyama, M. (2002) The p75 receptor transduces the signal from myelin-associated glycoprotein to Rho. J. Cell Biol. 157, 565-570
- Tanaka, H., Yamashita, T., Asada, M., Mizutani, S., Yoshikawa, H. & Tohyama, M. (2002) Cytoplasmic p21Cip1/WAF1 regulates neurite remodeling by inhibiting Rho-kinase activity. J. Cell Biol. 158, 321-329.
- Yamashita, T., Tucker, K. L. & Barde, Y.A. (1999) Neurotrophin binding to the p75 receptor modulates Rho activity and axonal outgrowth. Neuron 24, 585-593.
