マラリア免疫学
Malaria Immunology
TEL 03-6409-2219
FAX 03-6409-2221
概要
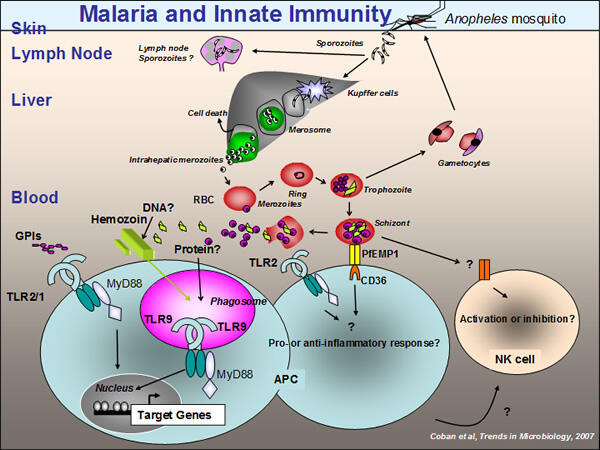
Malaria is responsible for the deaths of over a million people each year. Approximately half the world's population is at potential risk of the disease, which infects 300 million people annually. The Plasmodium parasites responsible for the disease have a complex life cycle which involves both a vertebrate (mammal) and invertebrate (Anopheles mosquito) host. The parasites have evolved many strategies that allow them to evade the host immune system (Figure 1).
The innate immune system is a highly specialized first line defense system against many infectious diseases, including malaria. However, the role of innate immunity in response to Plasmodium parasites is still unclear (Figure 1). We aim to better understand the role of host innate immune responses to malaria parasites by using mouse malaria models and genetically-modified mice lacking a variety of genes important for the innate immune system. We believe our studies will give new insights into the interactions between malaria parasites and the host innate immune response, and will enable us to develop new strategies such as new drugs or vaccines to combat this disease.
We aim to develop novel vaccines against malaria based on novel vaccination techniques and/or novel adjuvants. The development of a DNA vaccine is one the promising strategy for stimulating immunity against malaria. To improve the immunogenicity of such DNA vaccines in higher primates and humans, we have investigated molecules that may be used as novel adjuvants for improving vaccine immunogenicity.
We are also undertaking extensive studies to develop hemozoin, a malarial metabolite, as a new endogenous vaccine adjuvant against malaria and as an exogenous vaccine adjuvant for many protein antigens as well as in allergy models. We are also interested in the role of the innate immune system on hemozoin's adjuvant properties.
主任研究者
Cevayir Coban 教授

研究内容
Immunology and Parasitology, Immune responses to Plasmodium parasites, Vaccine development
学歴
| 1994 | MD, Hacettepe University Medical School, Ankara, TURKEY |
|---|---|
| 1998 | Degree in Clinical Microbiology, Ankara Numune Education and Training Hospital, Ankara, TURKEY |
職歴
| 1998 | Visiting Fellow, FDA/CBER (Food and Drug Administration), Bethesda, MD, USA (-1999) |
|---|---|
| 2000 | Postdoctoral Fellow, Johns Hopkins University School of Public Health, Malaria Research Institute, Baltimore, MD, USA (-2003) |
| 2003 | Postdoctoral Fellow, The 21st Century Center of Excellence (COE) Program, Research Institute for Microbial Diseases, Osaka University, JAPAN (-2006) |
| 2006 | Specially appointed Assistant Professor, The 21st Century Center of Excellence (COE) Program, Research Institute for Microbial Diseases, Osaka University, JAPAN (-2008) |
| 2008 | Assistant Professor, Laboratory of Host Defense, Immunology Frontier Research Center, World Premier Institute for Immunology, Osaka University, JAPAN (-2010) |
| 2010 | Associate Professor, Immunology Frontier Research Center, World Premier Institute for Immunology, Osaka University, JAPAN (-2015) |
| 2015 | Professor, Osaka University, JAPAN |
| 2019 | Specially Appointed Professor, Osaka University, JAPAN |
| 2019 | Professor, the Institute of Medical Science, the University of Tokyo, JAPAN |
メンバー
-
Cevayir Coban Professor
ccobanims.u-tokyo.ac.jp
業績
論文
-
Coban C, Lee MSJ, Ishii KJ. Tissue-specific immunopathology during malaria infection. Nature Reviews Immunology, 2018 doi:10.1038/nri.2017.138
- Lee MSJ, Maruyama K, Fujita Y, Konishi A, Lelliott PM, Itagaki S, Horii T, Lin JW, Khan SM, Kuroda E, Akira S, Ishii KJ, Coban C. Plasmodium products persist in the bone marrow and promote chronic bone loss. Science Immunology, 2017, June 2; 2 (12), pii: eaam8093. DOI: 10.1126/sciimmunol.aam8093.
-
Lelliott PM, Coban C. IFN-γ protects hepatocytes against Plasmodium vivax infection via LAP-like degradation of sporozoites. Commentary, Proc Natl Acad Sci U S A, 2016, Jun 10. pii: 201607007.
- Lee MSJ, Igari Y, Tsukui T, Ishii KJ, Coban C. Current status of synthetic hemozoin adjuvant: A preliminary safety evaluation. Review, Vaccine, 2016, Apr 19; 34(18):2055-61. doi:10.1016/j.vaccine.2016.02.064.
- Zhao H, Aoshi T, Kawai S, Mori Y, Konishi A, Ozkan M, Fujita Y, Haseda Y, Shimizu M, Kohyama M, Kobiyama K, Eto K, Nabekura J, Horii T, Ishino T, Yuda M, Hemmi H, Kaisho T, Akira S, Kinoshita M, Tohyama K, Yoshioka Y, Ishii KJ, Coban C Olfactory Plays a Key Role in Spatiotemporal Pathogenesis of Cerebral Malaria. Cell Host Microbe, 2014, 15(5): 551-63.
-
Zhao H, Konishi A, Fujita Y, Yagi M, Ohata K, Aoshi T, Itagaki S, Sato S, Narita H, Abdelgelil NH, Inoue M, Culleton R, Kaneko O, Nakagawa A, Horii T, Akira S, Ishii KJ, Coban C. Lipocalin 2 bolsters innate and adaptive immune responses to blood-stage malaria infection by reinforcing host iron metabolism. Cell Host Microbe, 2012, 12(5):705-16. doi: 10.1016/j.chom.2012.10.010.PMID: 23159059.
- Coban C, Igari Y, Yagi M, Reimer T, Koyama S, Aoshi T, Ohata K, Tsukui T, Takeshita F, Sakurai K, Ikegami T, Nakagawa A, Horii T, Nuñez G, Ishii KJ, Akira S. Immunogenicity of Whole Parasite Vaccines Against Plasmodium falciparum Involves Malarial Hemozoin and Host TLR9. Cell Host and Microbe, 2010, 7(1):50-61.
-
Coban C, Ishii KJ, Horii T, Akira S. Manipulation of host innate immune responses by the malaria parasite. Review, Trends in Microbiology, 2007, 15(6): 271-278.
- Coban C, Ishii KJ, Uematsu S, Arisue N, Sato S, Yamamoto M, Kawai T, Takeuchi O, Hisaeda H, Horii T, Akira S. Pathological role of Toll-like receptor signaling on cerebral malaria. International Immunology, 2007, 19(1):67-79.
- Coban C, Ishii KJ, Gursel M, Klinman DM, Kumar N. Effect of plasmid backbone modification by different human CpG motifs on the immunogenicity of DNA vaccine vectors. Journal of Leukocyte Biology, 2005, 78(3):647-55.
-
Coban C, Ishii KJ, Kawai T, Hemmi H, Sato S, Uematsu S, Yamamoto M, Takeuchi O, Itagaki S, Kumar N, Horii T, Akira S. Toll-like receptor 9 mediates innate immune activation by the malaria pigment hemozoin. Journal of Experimental Medicine, 2005, 201(1):19-25.
- Coban C, Philipp MT, Purcell JE, Keister DB, Okulate M, Martin DS, Kumar N. Induction of Plasmodium falciparum Transmission Blocking Antibodies in Nonhuman Primates by a Combination of DNA and Protein Immunizations. Infection and Immunity, 2004, 72(1):253-259.
